
OEM vs ODM vs Private Label: Which Model Fits You?
VEILTA
04/11/2026
In skincare manufacturing, OEM, ODM, and private label describe three different relationships between a brand and a factory. OEM means you own the formula and the factory makes it. ODM means the factory owns the formula and makes it for you. Private label means you buy an existing product off the shelf and put your brand on it.
That's the 30-second version. The longer version matters because choosing the wrong model for your stage of business is one of the most common — and most expensive — mistakes new skincare brands make.
The three models defined
OEM: Original Equipment Manufacturer
In a strict OEM arrangement, the brand brings the formula. You — or your R&D team — have developed a product specification: ingredient list, concentrations, texture profile, pH target. You're hiring a factory to manufacture it at scale.
The factory provides production equipment, raw material sourcing, in-process quality control, filling, sealing, packaging, and batch documentation. The brand retains formula ownership (documented in contract), exclusivity (the factory cannot sell the same formula to other brands), and full control over ingredient selection and product differentiation.
OEM is expensive to start. You need a formula before you need a factory — which means either hiring a cosmetic chemist, working with an R&D lab, or having in-house formulation capability. For a skincare patch, a custom formula development cycle typically runs 3–6 months before production begins. MOQs start higher because the factory is running dedicated production setups for a single client. For hydrogel acne patches specifically, OEM MOQs at most Chinese contract manufacturers start around 50,000 units, because the minimum viable batch size for the mixing and filling equipment is set by physics, not negotiation.
The payoff is genuine product differentiation. If your formula performs better than competitors', that advantage is yours and protected by contract. Before committing to any factory for OEM production, the vetting process deserves its own checklist — China Skincare Manufacturer Audit: 12 Questions B2B Buyers Always Forget covers the questions that reveal whether a manufacturer can actually deliver on a custom formula.
ODM: Original Design Manufacturer
ODM flips the model. The factory has developed a catalog of existing formulas — hydrocolloid acne patches at different active levels, hydrogel eye patches with various peptide combinations, facial sheet masks in multiple base materials. You select from that catalog, specify your branding and packaging, and the factory produces it under your label.
The brand gets a validated, tested formula with stability and safety data already completed, faster time to market with no formula development phase, and lower entry cost. The factory retains IP ownership of the base formula and the right to sell that same formula to other brands — unless exclusivity is negotiated separately.
ODM is how most new brands actually enter the market, though many won't admit it publicly. There's nothing wrong with it. The differentiation lives in branding, packaging, positioning, and the customer relationship — not necessarily in a proprietary formula. Plenty of successful brands are built entirely on ODM manufacturing.
The caveat: ODM exclusivity clauses are not standard. If you want to be the only brand your manufacturer supplies with a particular formula in your market, you need to negotiate that explicitly, pay a premium for it, and get it in writing — with a geographic scope and duration specified.
Private Label
Private label is sometimes used as a synonym for ODM, but in its most precise form it means buying a finished, packaged product that the factory already produces in bulk, then applying your own label to it.
The distinction from ODM: with ODM, there's usually at least some customization — your packaging design, your label, minor formula tweaks. With pure private label, you're buying something genuinely off the shelf. The factory may even have standard packaging that you simply put your logo on.
Private label has the lowest barrier to entry and the highest speed to market — as little as one to three weeks from order to finished goods. It also has the lowest differentiation. If four brands are all selling the same factory's standard hydrogel eye patch in the same packaging format, the competition becomes entirely about price, marketing, and distribution — not product quality. For a practical walkthrough of the full private label launch process for eye patches specifically, see How to Launch a Private Label Eye Patch in 90 Days.
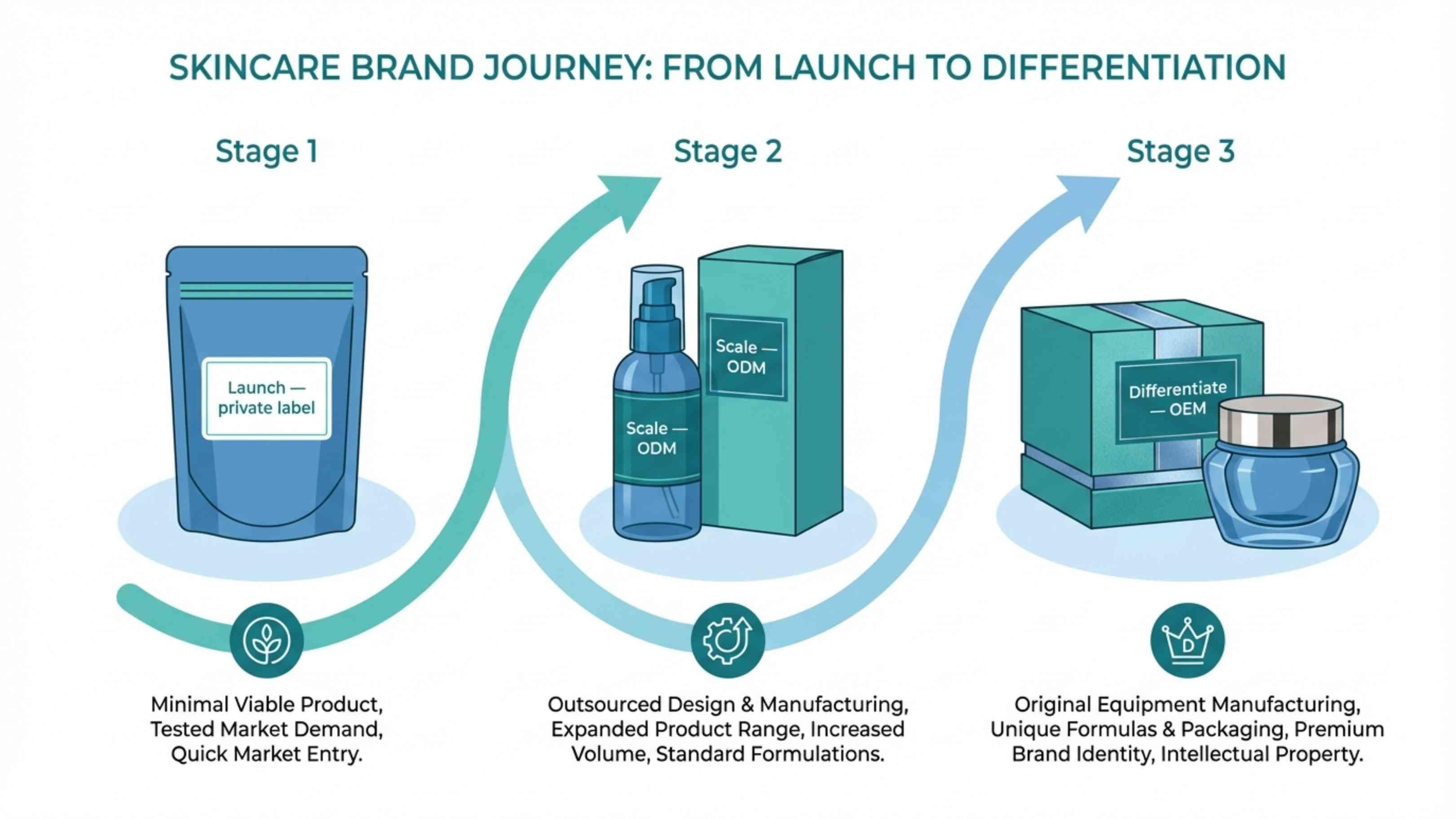
Use this as a starting framework. The right model depends on where your brand is now — not where you want to be in three years.
How the three models compare
| Factor | OEM | ODM | Private Label |
|---|---|---|---|
| Formula ownership | Brand | Factory | Factory |
| Customization level | Full | Partial | Minimal |
| Typical MOQ (patches) | 50,000+ units | 10,000–30,000 units | 3,000–10,000 units |
| Development time | 3–6 months | 4–8 weeks | 1–3 weeks |
| Formula exclusivity | Standard | Negotiable | Rarely available |
| Upfront investment | High | Medium | Low |
| Product differentiation | High | Medium | Low |
| IP risk if you switch factories | Low (you own the formula) | Medium (factory owns formula) | Low (no formula to lose) |
| Best suited for | Established brands with R&D capability | Brands launching new categories | Market testing, low-volume launches |
According to a 2023 industry survey by Mintel on beauty brand sourcing strategies, over 60% of indie skincare brands launching their first product used private label or ODM manufacturing — with fewer than 15% starting with a fully custom OEM formula. The shift to OEM typically happens after brands reach sustained monthly revenue that justifies the development investment.
VEILTA's perspective: The majority of first inquiries we receive are framed as OEM requests, but when we get into the conversation, the actual requirement is ODM or private label — the brand hasn't developed a formula yet and just wants custom packaging. That's completely workable, but it changes the timeline and MOQ conversation significantly. Knowing which model you're actually pursuing before the first call saves everyone a lot of back-and-forth.
Which model fits your situation: a decision framework
The simplest way to figure out where you stand is to answer four questions in order. The answers will route you to the right model more reliably than any general recommendation.
Question 1: Do you have a finished formula specification right now? If yes — ingredient list, concentrations, INCI names, target pH and viscosity — you can pursue OEM. If no, you're starting from ODM or private label regardless of what you call it.
Question 2: Does your product differentiation depend on the formula being unique? If your brand story is built on a specific active ingredient combination, a proprietary extraction process, or a patented delivery mechanism, formula exclusivity matters. That points toward OEM or ODM-with-exclusivity. If your differentiation is branding, design, and positioning, the formula doesn't need to be unique and ODM works fine.
Question 3: How much time do you have before launch? If you need to be on shelf or live on DTC within 8–12 weeks, OEM is off the table — the development cycle alone runs 3–6 months before a single unit is produced. ODM can realistically hit 8–12 weeks. Private label can hit 2–4 weeks.
Question 4: What is your realistic first-order volume? If you can commit to 50,000 units or more of a single SKU, OEM is accessible at most factories. Between 10,000 and 30,000 units, ODM is the realistic option. Below 10,000 units, private label is the only model most manufacturers will quote.
The path most brands actually follow: private label or ODM to validate market demand, then OEM once the sales data justifies custom development. There's no shame in starting at the bottom of the model stack — it's faster, cheaper, and lets you find out if the market actually wants the product before you spend on proprietary formulation.
A note on terminology confusion in Chinese manufacturing
In Chinese manufacturing and export contexts, "OEM" is used as an umbrella term for contract manufacturing of any kind — covering what Western B2B buyers would distinguish as OEM, ODM, or private label. When a factory says "we offer OEM services," they almost certainly mean they can do all three.
The label doesn't matter much. What matters is getting specific answers to four questions:
- Who owns the formula after production?
- What level of customization is available, and what are the minimum order requirements for each level?
- What are the exclusivity terms — geographic scope, duration, and price?
- What is the lead time and MOQ for each option?
A factory that answers these questions clearly is a factory you can work with. One that deflects or gives vague answers is one to approach cautiously.
IP protection: what actually happens at contract expiry
This is where the model choice has consequences that brands don't always think through until it's too late.
In an OEM arrangement, you own the formula. But ownership is only as good as the contract that documents it. The manufacturing agreement should explicitly state that all formula documentation — ingredient specifications, INCI names, concentrations, the manufacturing process description, and all stability and safety testing data — transfers to you if the relationship ends. It should also include a clause preventing the factory from reverse-engineering or adapting your formula for other clients.
Without that clause, a factory may argue that the production know-how is their proprietary process, even if the base formula originated with you. This scenario plays out more often than it should: a brand commissions OEM development, spends six months and significant budget on formulation, then decides to switch manufacturers after a quality issue — and discovers the factory is reluctant to hand over the batch records and production documentation. Negotiate data transfer terms before production begins. For a broader look at what GMP certification actually covers — and what to verify at the factory level before any contract is signed — see GMP Cosmetics Manufacturing: What the Certification Actually Covers.
In an ODM arrangement, the formula belongs to the factory. If you've contributed to further development — funded stability testing on a new active combination, for example — negotiate whether that contribution earns you any rights to the modified formula, or whether you're simply paying for a service that the factory then owns. Most factories will say the latter unless you argue otherwise in writing.
In private label, there's usually nothing to negotiate on the formula because there's no formula IP to speak of — you're buying a commodity product. Your IP is your brand, your trademark, your packaging design, and your trade dress. Make sure those are registered in your target markets before launch, regardless of which manufacturing model you use. That protection costs far less than defending an unregistered brand against a copycat.
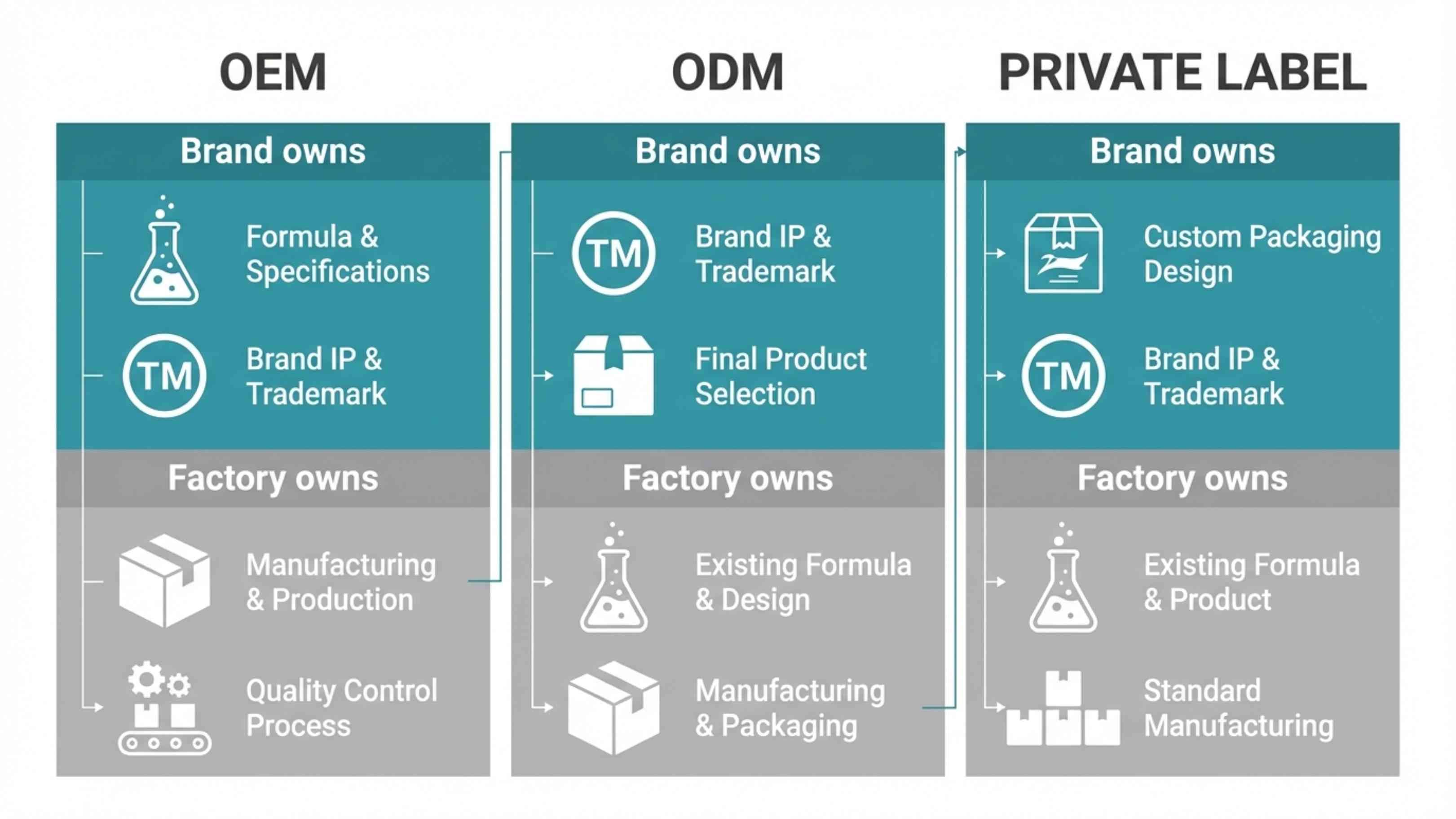
IP ownership is the single most important structural difference between the three models. The contract is where ownership is defined — a handshake or a verbal agreement is not sufficient.
Running multiple models simultaneously
Many brands run all three models across different product lines at the same time — and this is often the most practical approach.
A realistic example: a brand has two hero products that are genuine OEM formulations with strong IP protection, five supporting products developed through ODM (fast to market, tested by the factory, exclusivity negotiated for their primary market), and a handful of private label accessories and travel-size items used for bundles, gift-with-purchase promotions, and seasonal campaigns.
The hero products carry the brand's scientific credibility. The ODM products fill out the range and let the brand compete across price points without the overhead of custom development on every SKU. The private label items serve tactical marketing needs without consuming R&D budget.
This portfolio approach is well-suited to brands that are scaling — you don't have to commit everything to one model and live with its constraints across your entire product line.
A blended portfolio: OEM for hero differentiation, ODM for range breadth, private label for tactical volume. Most scaling brands eventually run all three.
FAQ
What is the difference between OEM and ODM in skincare?
OEM means the brand provides the formula and the factory manufactures it — formula ownership sits with the brand. ODM means the factory developed the formula and the brand sells it under their own name — formula ownership sits with the factory. The practical difference shows up in three places: who pays for R&D (the brand in OEM, the factory in ODM), who can sell the formula to other clients (only the brand's licensee in OEM, anyone the factory chooses in ODM unless exclusivity is agreed), and how long it takes to get to market (months longer for OEM due to development time).
What is private label in skincare manufacturing?
Private label means buying a factory's existing finished product and selling it under your own brand. The formula is pre-developed, pre-tested, and often already in production for multiple other buyers. You typically get to specify your packaging design and label copy, but the formulation itself isn't exclusive. Speed to market is the main advantage — you're not waiting for R&D or stability testing because that work is already done. The tradeoff is that competitors can source the same or very similar product from the same factory.
Which model has the lowest MOQ?
Private label, consistently. The factory has materials stocked and production lines already configured for the product, so small runs are viable. ODM typically requires 10,000–30,000 units for skincare patches because some setup is involved. OEM starts at 50,000 units or higher at most contract manufacturers, because running a dedicated batch for a single client formula only makes economic sense above a threshold volume. These numbers vary by product type — microneedle patches have higher minimums than standard hydrocolloid patches regardless of model.
Can I switch from private label to OEM later?
Yes, and this is a well-worn path. Most brands start with private label or ODM to prove market demand, then commission OEM formula development once they have sales data showing the category is viable and the volume to justify the investment. The transition usually takes 6–9 months from briefing a chemist to having a production-ready OEM formula. Plan that timeline before you need it — starting the OEM process while you're still running on private label sales is far smoother than waiting until you've already scaled and are scrambling to differentiate.
Does ODM mean the factory owns my formula?
In a standard ODM arrangement, yes — the factory created the formula and retains ownership. What you own is your brand, your packaging design, and your market positioning. You can negotiate exclusivity (the factory won't sell the same formula to your direct competitors in your market), but that requires explicit negotiation, a written clause in the manufacturing agreement, and usually a premium MOQ or price. Geographic scope and duration both need to be defined — "exclusive" without those parameters is not meaningful protection.
What happens to my OEM formula if I switch manufacturers?
This depends entirely on what your manufacturing contract says. In a well-drafted OEM agreement, all formula documentation — full ingredient specification, INCI names, concentrations, manufacturing process, stability data — belongs to you and must be transferred on request. In a poorly drafted agreement (or a handshake arrangement), the factory may claim that the production process is their know-how and resist handing over documentation. Negotiate data transfer terms explicitly before production starts. It's a short conversation to have upfront and a very long one to have when you're trying to leave. If you're sourcing pimple patches specifically, OEM Pimple Patches: Sourcing Guide for Brand Owners includes a section on contract terms worth reviewing before the first factory call.
What does "full OEM service" mean when Chinese manufacturers use it?
It usually means turnkey contract manufacturing — the factory handles R&D, raw material sourcing, production, filling, and packaging. Whether the underlying arrangement is technically OEM (your formula), ODM (their formula), or private label (their existing product) varies by client. The phrase "full OEM" in Chinese manufacturing exports is essentially synonymous with "we handle everything end-to-end." What matters is not the label but the specific answers to: who owns the formula, what customization is available, and what the exclusivity terms are.
Is private label the same as white label?
Functionally the same in most skincare manufacturing contexts. White label is sometimes used to describe a more generic version — completely unbranded product that multiple buyers purchase identically — while private label implies slightly more customization: your own packaging design, maybe a fragrance variant or different label claims. In practice, most factories in China use both terms interchangeably and mean the same thing: existing formula, your branding. If the distinction matters for your use case, just ask the factory directly what level of customization is included in what they're offering.
