
OEM Pimple Patches: Sourcing Guide for Brand Owners
VEILTA
03/22/2026
Sourcing pimple patches for your brand? Veilta manufactures hydrocolloid acne patches, private label skincare, and custom formulations for brands across the US, EU, and Southeast Asia — out of Zhongshan, China, with GMP certification. Request a free sample →

Five pimple patch formats available for private label at Veilta — from classic hydrocolloid discs to dissolving microneedle patches.
The acne patch category looks simple from the outside. Small sticker, hydrocolloid material, apply and leave overnight. The product is easy to explain to consumers. Getting from "we want to launch one" to "units are in our warehouse" is a different story — and the gap between those two points is where most brands lose time.
This guide covers the sourcing process from the beginning: what the product actually does, how OEM and ODM differ, what formats are available, what the regulatory requirements are, and the questions worth asking before you commit to a manufacturer.
Quick summary
- The global acne patch market was valued at over USD 1.2 billion in 2023, forecast to grow at 8.5% CAGR through 2030 (Grand View Research, 2024)
- OEM (custom formula) requires 10,000+ unit MOQ and 60–90 day lead time; ODM (stock formula) starts at 3,000 units and 30–45 days
- Five main formats: classic hydrocolloid, microneedle, colored/shaped, medicated, and ultra-thin
- Compliance requirements differ by market — US FDA listing, EU CPNP notification, and ISO 22716 GMP are the baseline
What are OEM pimple patches?
OEM pimple patches — also called private label acne patches or hydrocolloid blemish stickers — are manufactured by a contract factory and sold under your brand's label. The brand owns the positioning, packaging, and retail relationship. The factory handles production.
For brands entering the acne care category, OEM is often the most practical starting point. You don't need a lab or a production line. You need a factory that can make the product to spec, at your required volume, with documentation your target market will accept.
Market context: the global acne patch category was valued at over USD 1.2 billion in 2023 and is forecast to grow at 8.5% CAGR through 2030, with demand concentrated in Gen Z and the ongoing K-beauty influence on Western skincare (Grand View Research, 2024).
How do pimple patches work?
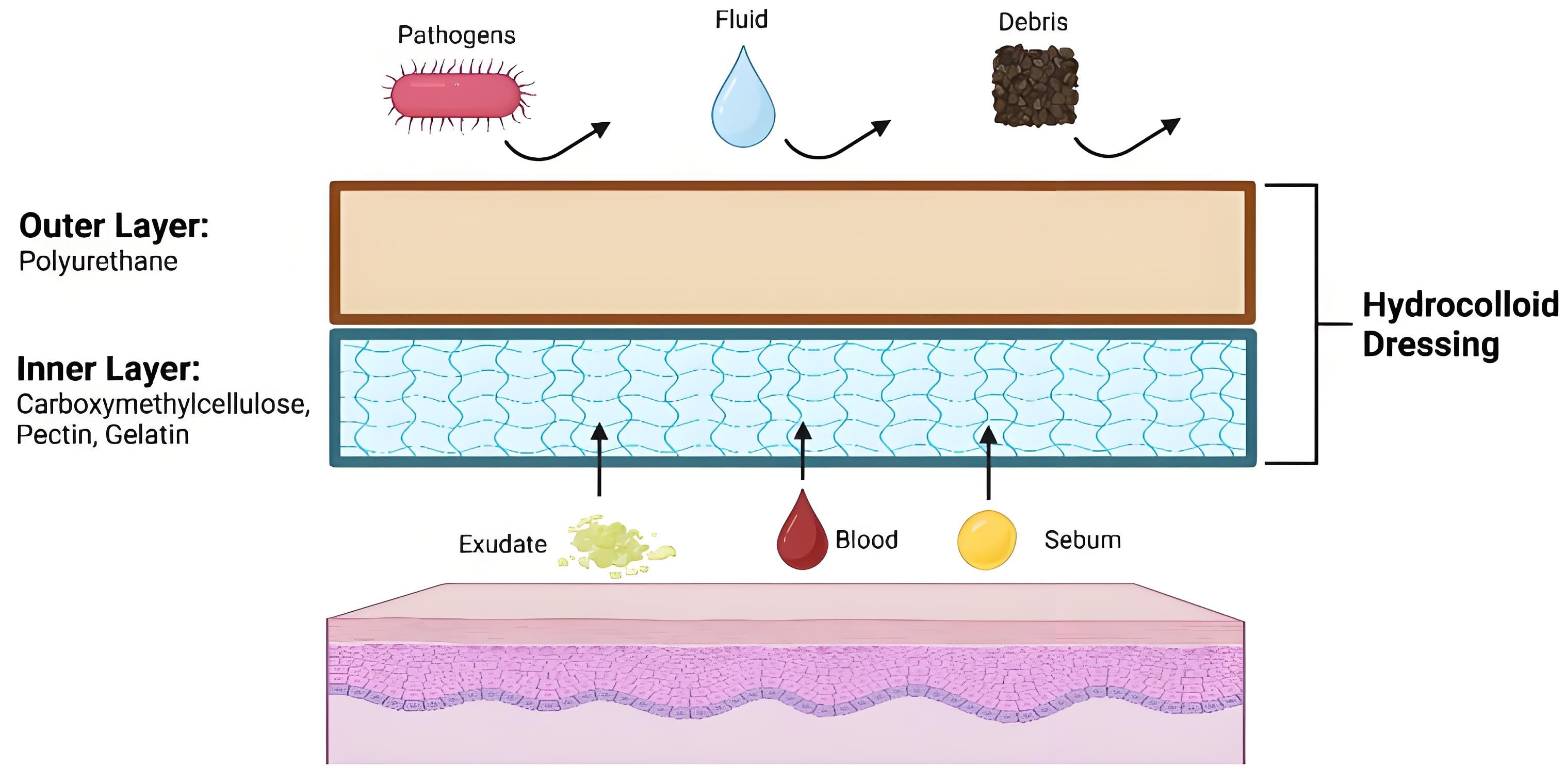
The active material is hydrocolloid — a moisture-absorbing polymer originally developed for wound dressings in clinical settings. When a patch sits on a blemish, it pulls fluid (pus, sebum, exudate) out of the skin through osmotic pressure, flattens the spot, and puts a physical barrier between the wound and outside bacteria.
Clinically, hydrocolloid dressings reduce healing time by up to 40% compared to leaving a wound exposed (Journal of Wound Care, 2021). That's the mechanism behind every pimple patch on the market — the format differences come down to thickness, active ingredients, and whether there are microneedles involved.
From a brand perspective, this is one of the more self-explanatory product categories. The mechanism is simple, the visual result (patch turns white as it absorbs) is satisfying, and the repeat purchase rate is high.
How hydrocolloid works: the patch draws fluid out of the blemish through osmotic pressure and turns white as it fills — a visible signal that it's working.
OEM vs ODM: which model fits your situation?
These two terms get used interchangeably but they're different contracts.
| OEM | ODM | |
|---|---|---|
| Formula | Your custom formulation | Factory's existing formula |
| Packaging | Your full design | Your logo on the factory's standard pack |
| MOQ | 10,000+ units typically | 3,000–5,000 units typically |
| Lead time | 60–90 days | 30–45 days |
| Best for | Brands with R&D and volume | Startups, new SKU testing |
| IP ownership | Brand owns the formula | Factory retains or shared |
Most brands launching in acne care start with ODM. It's faster, the MOQ is lower, and you can validate whether there's real demand before committing to a custom formulation. Once a SKU is generating consistent reorders, that's the point where moving to OEM — with a formula you own — makes financial sense.
Pimple patch formats available for private label
Classic hydrocolloid patches
The core of the category. Transparent or white disc patches, available in 8mm, 10mm, and 12mm diameters. These are the patches most consumers are familiar with, and they're the most accessible price point for mass market or pharmacy-channel brands. MOQ at Veilta starts at 5,000 units.
Microneedle patches
Dissolving microneedles carry actives — salicylic acid, niacinamide, hyaluronic acid — directly into the blemish rather than just pulling fluid out. The retail price point is typically 3–5x that of classic patches. Search interest grew 65% year-over-year in 2024 (Google Trends), which makes this the highest-growth segment in the category right now.
Colored and shaped patches
Star shapes, pastel colors, visible patches designed to be worn in public. The Gen Z and teen segment drove this format. The product logic is different — it's about normalizing acne treatment and generating social media content rather than hiding blemishes. Works well for DTC brands with strong Instagram or TikTok presence.
Medicated patches
Patches with regulated actives: salicylic acid at 0.5–2%, tea tree oil, centella asiatica. These are effective, but they introduce regulatory complexity. In the US, salicylic acid above 0.5% puts you in OTC drug territory (FDA 510(k) pathway). In the EU, you're looking at CE marking and specific dossier requirements. Veilta handles the documentation side of this — it's manageable, just plan for longer lead times.
Invisible / ultra-thin patches
Matte finish, designed to disappear under makeup. Daytime positioning. Premium segment. The manufacturing tolerances are tighter, which is reflected in the unit cost.
How to apply pimple patches — usage guide for your packaging and listings
Copy this directly for packaging inserts or product pages.
Step 1 — Cleanse. Wash with a gentle cleanser and dry completely. Moisture left on the skin reduces how well the patch sticks.
Step 2 — No product on the target spot. Don't apply moisturizer, serum, or anything else over the blemish first. The hydrocolloid needs bare skin contact to absorb properly.
Step 3 — Apply. Peel from the liner, press onto the blemish for 5–10 seconds, smooth down the edges. No air bubbles.
Step 4 — Leave it. 6–8 hours minimum, overnight if possible. The patch turns white when it's full — that's when you replace it.
Step 5 — Resume your routine. Once the patch is on, apply moisturizer and SPF to the rest of your face. Don't put anything directly over the patch.
Want to sample these formats before committing to an order? Request samples from Veilta →
What to ask an OEM manufacturer before placing a first order
Most sourcing mistakes happen before production starts — not during it. These are the five questions worth asking any manufacturer before you sign off on a first order.
1. Can you share COAs and stability test results for the formula I'm buying? A Certificate of Analysis shows the exact ingredient composition and test results for a specific batch. Stability test data confirms the formula performs as intended over its shelf life. Reputable manufacturers have these on file and share them without hesitation. If a factory is reluctant to provide COAs, that's a clear signal.
2. Who owns the formula after production — me or you? Under ODM, the factory typically retains formula ownership. Under OEM, the brand owns it. The distinction matters when you want to switch manufacturers or scale to a different factory later. Get the IP terms in writing before you pay any deposit.
3. What does your pre-shipment inspection process look like? Find out whether the factory runs internal QC against a signed golden sample, and whether they support third-party inspection (SGS, Bureau Veritas, or equivalent). For a first order with a new supplier, independent inspection is worth the cost — typically $200–400 — against the risk of receiving a non-conforming shipment.
4. What compliance documentation do you provide for my target market? For the US: FDA facility registration confirmation and ISO 22716 GMP certificate. For the EU: CPSR-ready formula documentation and support for CPNP notification. For Southeast Asia: BPOM, HSA, or NPRA documentation depending on the country. A factory that can't answer this question specifically is not ready to serve export markets.
5. What are your payment terms and what happens if production doesn't match the approved sample? Standard terms are 30% deposit upfront, 70% balance before shipment. More important: establish in writing what the remediation process is if bulk production deviates from the golden sample — rework, replacement, or credit. This protects you without requiring litigation.
Frequently asked questions
What's the MOQ for OEM pimple patches?
ODM orders start at 3,000–5,000 units. Fully custom OEM formulations typically require 10,000 units minimum. If your initial volume is lower, ODM is the more practical option. Talk to us about your specific situation →
How long does production take?
ODM: 30–45 days from approved artwork. OEM with a custom formula: 60–90 days, which includes stability testing. Rush production is available at additional cost.
What certifications does a manufacturer need for the US and EU?
For the US: FDA facility registration (mandatory under MoCRA 2022) and ISO 22716 GMP certification. For the EU: ISO 22716 GMP, plus an appointed EU Responsible Person and CPNP notification before selling. Both processes should start in parallel with sampling, not after production. Veilta's facility is GMP-certified and compliant with EU Cosmetics Regulation (EC) No 1223/2009. We support clients through FDA, CE, CPNP, and CPON registration.
Can I add custom active ingredients?
Yes. The most common additions are salicylic acid, niacinamide, centella asiatica extract, and tea tree oil. Note that salicylic acid above 2% triggers different regulatory classifications depending on the market. Our regulatory team will flag this early so it doesn't delay your timeline.
What packaging formats do you offer?
Blister cards, resealable pouches, tin cases, and retail-ready boxes. We provide dieline files and can coordinate with your designer or handle the artwork internally.
Should pimple patches go on before or after moisturizer?
Before. Always on clean, dry skin. Applying over moisturizer stops the hydrocolloid from adhering to the skin surface, which means it can't absorb anything.
Is toner okay before patching?
A water-based toner is fine, as long as it's fully absorbed before you apply the patch. Oil-based or emollient toners will interfere with adhesion.
How long do you leave a patch on?
6–8 hours minimum. Overnight is better. When the patch turns fully white and opaque, it's saturated — replace it rather than leaving it on.
Veilta manufacturing at a glance

Veilta's GMP-certified production facility in Zhongshan, Guangdong. ISO 22716 compliant, with in-house stability testing and regulatory documentation for US, EU, and Southeast Asian markets.
| Location | Zhongshan, Guangdong, China |
| Certifications | GMP, ISO 22716, EU Cosmetics Reg. compliant |
| R&D | In-house formulation team, stability and safety testing on site |
| MOQ | From 3,000 units (ODM) |
| Lead time | 30–90 days depending on scope |
| Markets served | US, EU, UK, Australia, Southeast Asia, Middle East |
| Support | Regulatory docs, artwork review, logistics coordination |
Source your pimple patches with Veilta
The quickest way to evaluate what we make is to request a sample. We send factory overview documentation along with physical samples so you can assess quality and compliance in one step.
Or reach us directly at service@veilta.com
