
Pimple Patch Compliance: FDA, CPNP & BPOM Guide (2026)
VEILTA
03/19/2026
You've locked in your manufacturer, signed off on the formula, and you're two weeks from placing your first bulk order.
Then someone asks: "Wait — is this actually compliant for the US market?"
It's the question that derails more launch timelines than any sourcing problem or packaging delay. Not because compliance is complicated, but because most brand owners only start thinking about it too late.
This guide covers what you actually need to know before selling pimple patches in the US, EU, UK, and beyond — including where the common mistakes happen and what each market genuinely requires versus what's just nice to have.
TL;DR
- Most hydrocolloid pimple patches are cosmetics, not drugs — but the moment you make acne treatment claims, the regulatory pathway changes completely.
- The US (FDA/MoCRA), EU (CPNP), and UK (SCPN) each require separate registrations. One notification does not cover all three.
- Selling in the EU without an appointed EU Responsible Person is illegal, regardless of where your brand is based.
- Indonesia (BPOM) and the Philippines (FDA Philippines) require 6–12 months of lead time — start registration in parallel with product development.
- "FDA certified" doesn't exist for cosmetics. It means registered, not approved.
First: Mandatory vs. Voluntary (and Why Most Brands Confuse the Two)
Not everything called a "certification" carries the same weight.
Some requirements are legally mandatory. You either meet them or you can't sell — period. Getting it wrong means customs seizures, forced recalls, fines, or worse. These aren't negotiable and they're not something you can sort out after the fact.
Others are voluntary certifications — cruelty-free seals, organic standards, dermatologist-tested claims. These can influence purchasing decisions in certain markets, but skipping them won't land you in legal trouble.
Most of this guide is about the mandatory stuff. The voluntary certifications get their own section at the end.
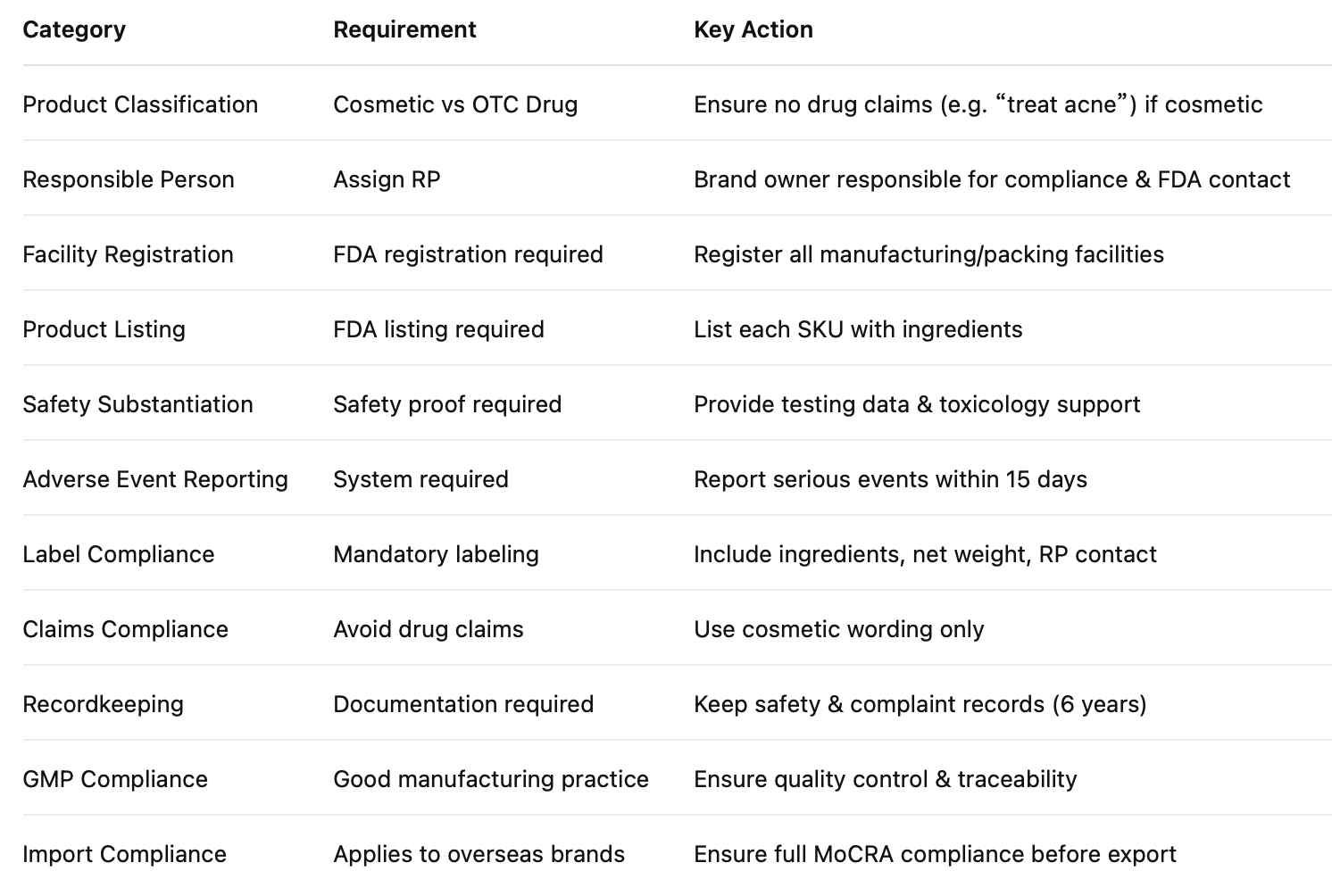
United States: FDA
The FDA regulates cosmetics under the Federal Food, Drug, and Cosmetic Act, which got a meaningful update in 2022 through the Modernization of Cosmetics Regulation Act (MoCRA). As of 2026, MoCRA is fully in effect.
Cosmetic or drug — and why this question matters more than you think
The most consequential decision you'll make for the US market isn't about ingredients or packaging. It's about classification.
A hydrocolloid patch is almost certainly a cosmetic. A patch making acne treatment claims may be an OTC drug. And those two pathways have almost nothing in common from a compliance standpoint.
The trigger is the claim on your label, not just what's in the formula. A patch that says "absorbs pimple fluid overnight" is a cosmetic. The same patch reworded as "treats acne" or "kills acne-causing bacteria" is potentially an OTC drug. Same product, completely different regulatory reality.
Salicylic acid makes this more complicated. The FDA recognizes it as an OTC active ingredient for acne at 0.5%–2% under 21 CFR Part 333. If your patch contains salicylic acid and you're making acne treatment claims, you're in drug territory — which means OTC Drug Monograph compliance, Drug Facts labeling on the box, a different facility registration category, and adverse event reporting obligations. If you want to use salicylic acid as a cosmetic ingredient without the OTC drug burden, keep your claims in clearly cosmetic territory and confirm your concentration stays within cosmetic-permitted limits.
When in doubt, get a regulatory opinion before you finalize your label copy. It's cheap insurance.

What MoCRA actually changed
Before MoCRA, FDA oversight of cosmetics was relatively light. The 2022 update introduced several requirements that are now standard:
Every facility that manufactures or processes cosmetics for the US market must be registered with the FDA — including your factory in China or Korea. Registration renews every two years. Each product must be listed in the FDA's Cosmetics Direct portal with a full ingredient list. Serious adverse events (hospitalizations, significant injury, disfigurement) must be reported to the FDA within 15 business days. And brands need to be able to demonstrate product safety — not through pre-market approval, but through safety documentation kept on file.
GMP regulations for cosmetics are also coming. They're not finalized yet as of 2026, but the direction is clear.
Before you sell in the US, confirm:
- Product classification confirmed (cosmetic or OTC drug), based on both ingredients and claims
- Your manufacturer's FDA facility registration is current
- Product listed in FDA Cosmetics Direct portal
- Label compliant with FPLA: product identity, net quantity, responsible party name and address, INCI ingredient list, country of origin
- If OTC drug: Drug Facts labeling, OTC monograph compliance, drug facility registration
- Adverse event reporting process in place
- Safety substantiation documentation on file
One more thing: "FDA certified" doesn't exist
If a manufacturer tells you their facility is "FDA certified," what they mean is that it's FDA registered. That's a mandatory legal baseline, not a quality endorsement. The FDA doesn't certify or approve cosmetic products before they go to market. Brands using "FDA approved" language on cosmetic packaging are making a claim that isn't accurate — and that can itself become a compliance issue.
European Union: CPNP
The EU has the most demanding cosmetics regulations in the world, governed by EU Cosmetics Regulation (EC) No 1223/2009. If you're planning to sell in Europe, this is the framework you'll spend the most time with.
The CPNP notification
Every cosmetic product must be notified on the Cosmetic Products Notification Portal (CPNP) before it enters the EU market. Not after — before. Each SKU gets its own notification. You get a CPNP reference number as confirmation, and without it, the product isn't legally on the market.
The notification can only be submitted by an EU Responsible Person. If your brand is based outside the EU, you cannot submit it yourself.
The EU Responsible Person
This is the requirement that catches most non-EU brands off guard.
To sell cosmetics in the EU, you need a legal entity based within the EU who takes on formal responsibility for your product's compliance. That entity can be your EU distributor or importer (if they're willing), a third-party compliance service provider (several companies do this as their core business), or your manufacturer if they have an EU legal entity.
The Responsible Person maintains your CPNP notification, keeps your Product Information File current, and is the contact point if authorities come asking. Expect to pay €300–1,000+ per year depending on the provider and how many SKUs you have.
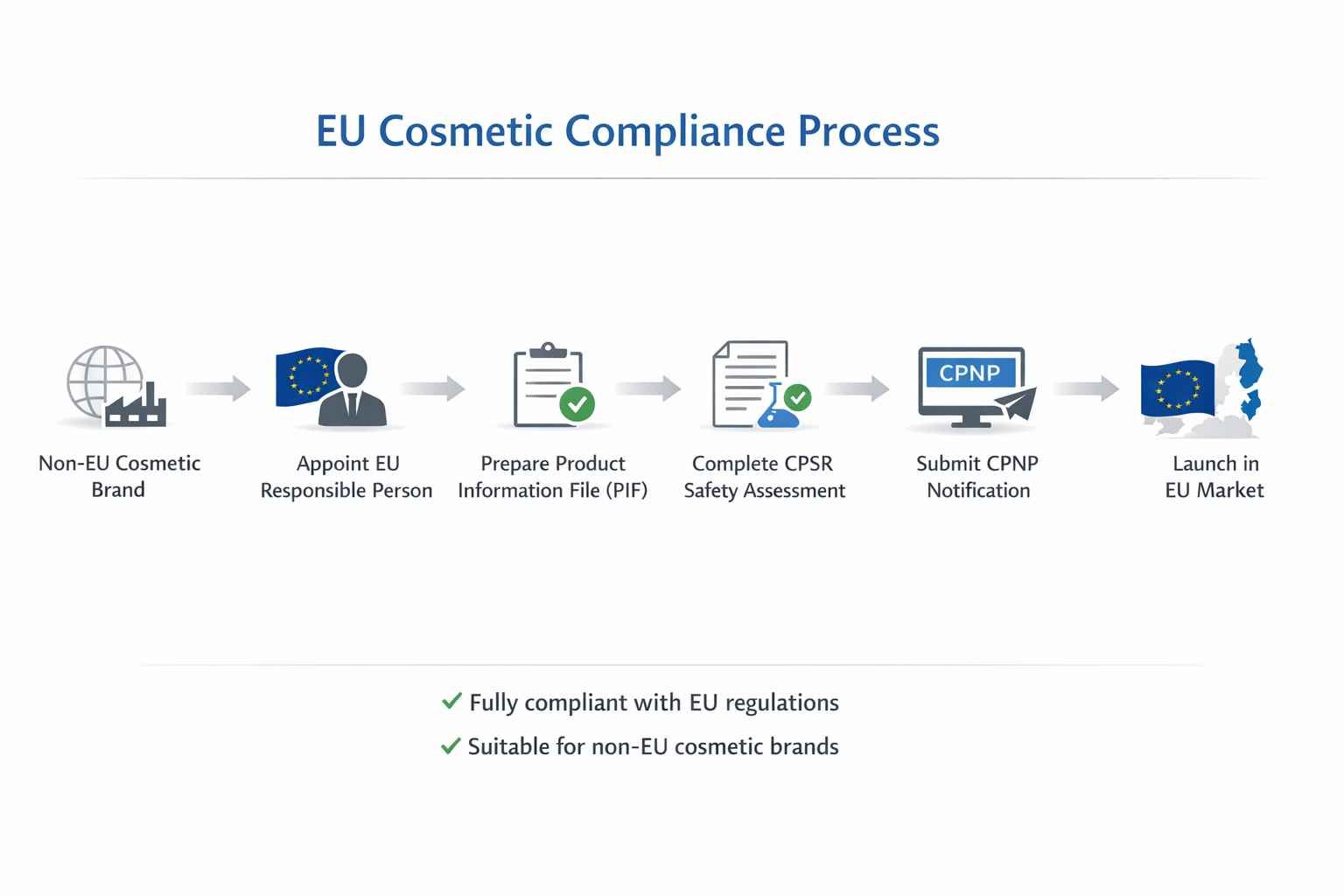
The Product Information File
Every EU cosmetic needs a PIF — a technical dossier that the Responsible Person maintains for at least 10 years after the last batch sells. It includes the complete formula with actual concentrations (not just the INCI list), manufacturing documentation, the cosmetic product safety report (CPSR), and evidence backing any claims on your packaging.
The CPSR is the most involved piece. It must be prepared and signed by a qualified safety assessor — typically a toxicologist with EU qualifications. Budget €500–1,500 per product.
Ingredient restrictions worth knowing
The EU restricts a long list of ingredients. For pimple patches specifically:
Salicylic acid is permitted in leave-on products (patches qualify as leave-on) up to 2.0%. Products containing it must carry a warning: "Contains Salicylic Acid. Not to be used on children under 3 years except under medical supervision." Tea tree oil has concentration limits depending on whether it's used as a fragrance or an active ingredient. And 26 fragrance allergens must be individually declared on the label if concentration exceeds 0.001% in leave-on products.
EU label requirements
Labels must be in the official language of each member state where you're selling — which in practice means a multi-language label if you're selling across multiple EU countries. Required elements include: the Responsible Person's name and address, country of origin, net content, period after opening (PAO) symbol or best-before date, batch number, INCI ingredient list, and any mandatory warnings.
United Kingdom: SCPN
Brexit created a separate UK regulatory framework. EU CPNP notifications don't cover the UK — you need a distinct registration.
The UK equivalent is the Submit Cosmetic Product Notifications (SCPN) portal, operated by the Office for Product Safety and Standards. Like the EU, you need a Responsible Person — but this one must be based in Great Britain (England, Scotland, or Wales). Northern Ireland is a separate situation: under the Windsor Framework, products sold there still follow EU rules, meaning CPNP notification applies.
The substantive requirements are similar to the EU — same PIF structure, same safety assessment approach, English-language labeling. But it's a separate process, a separate portal, and a separate Responsible Person. If you're targeting both EU and UK, budget for both.

Preparing to enter the EU or UK market? VEILTA provides full CPNP documentation support — including complete formula files, COAs, and safety assessment data — for brands sourcing through us. Talk to our team about your EU compliance requirements →
ISO 22716 GMP: The Manufacturing Standard
ISO 22716 is the international Good Manufacturing Practice standard for cosmetics. It's not a market access requirement in the same way as FDA registration or CPNP — but it's increasingly treated as one.
Major retailers (Sephora, Boots, Ulta) and EU distributors routinely require ISO 22716 certification as a condition of listing. Your manufacturer holding this certification means their processes have been independently audited — batch records, raw material traceability, in-process quality controls, the full picture.
From a practical standpoint: if you're planning retail distribution at any point, your manufacturer's ISO 22716 status is something you need to confirm upfront, not discover later.
CE Marking: When It Applies (And When It Doesn't)
CE marking is a medical device designation, not a cosmetic one. For the vast majority of private label pimple patch brands, it's not relevant.
Standard hydrocolloid patches sold with cosmetic positioning — "absorbs pimple fluid," "protects blemishes," "promotes clearer-looking skin" — are cosmetics in the EU. CPNP applies. CE marking does not.
CE marking becomes relevant only if your product crosses into medical device classification: wound healing claims, intended use following a medical procedure, or treatment of acne as a medical condition rather than a cosmetic concern. If you're specifically targeting wound care or medical aesthetics channels, that requires specialist regulatory expertise well beyond standard cosmetics compliance.
Stay in cosmetic claim territory and CE marking isn't something you need to think about.
Australia, Canada, and Southeast Asia
Australia
Standard cosmetic pimple patches sit outside the TGA (Therapeutic Goods Administration) framework. For most hydrocolloid patches with cosmetic positioning, compliance means AICIS (Australian Industrial Chemicals Introduction Scheme) compliance for any new chemical ingredients and standard cosmetic labeling under Australian Consumer Law. No pre-market registration required.
TGA gets involved only if you're making therapeutic claims — treating acne as a medical condition, for example. Avoid that language on packaging for Australia and you're in the simpler pathway.
Canada
Canada is relatively straightforward. Cosmetics are regulated by Health Canada under the Food and Drugs Act. Unlike most markets, notification is post-market — you file within 10 days of first sale, not before. The main requirements are Cosmetic Ingredient Hotlist compliance (Canada's restricted ingredients list), and bilingual labeling in English and French.
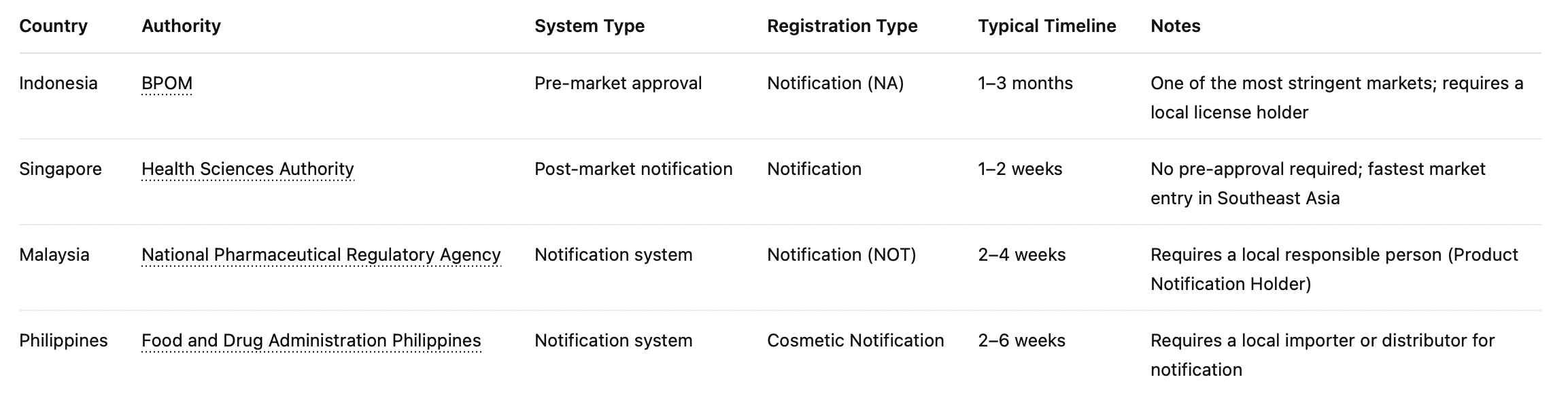
Southeast Asia
There's no ASEAN-wide cosmetics framework. Each country is its own registration process, its own regulatory body, and its own timeline:
| Country | Regulatory Body | Typical Timeline |
|---|---|---|
| Indonesia | BPOM | 3–6 months |
| Singapore | HSA (PRISM portal) | Several weeks |
| Malaysia | NPRA (QUEST portal) | Weeks to months |
| Thailand | FDA Thailand | Varies by product type |
| Philippines | FDA Philippines | 6–12 months |
| Vietnam | DAV | Varies |
Indonesia and the Philippines are the ones to plan around carefully. BPOM registration can take three to six months; Philippines FDA routinely takes six to twelve. If either market is on your roadmap, start the registration process in parallel with product development — not after your shipment is sitting in a warehouse.
Voluntary Certifications Worth Considering
Cruelty-free
Leaping Bunny (CCIC) and PETA's Beauty Without Bunnies are the two certifications consumers recognize most. Both require no animal testing at any stage of production or raw material sourcing, with annual audits. For brands targeting clean beauty retail — Sephora Clean, Space NK — cruelty-free certification is increasingly a listing requirement, not just a nice-to-have.
Vegan
Most standard hydrocolloid formulations are already vegan, but worth verifying — some formulations use gelatin, which is animal-derived. If your formula is vegan, getting the Vegan Society Trademark or PETA Vegan certification formalizes that claim with a recognized mark.
Dermatologist-tested
This one is worth understanding clearly: "dermatologist-tested" is a claim, not a third-party certification. To make it legitimately, you need documentation — typically a consumer use study conducted under dermatologist supervision and the dermatologist's written sign-off. Keep that on file. It carries weight in pharmacy channels and with consumers who have sensitive skin.
Organic/natural certifications
COSMOS and ECOCERT are the main standards here. Relevant if you've specifically formulated with certified organic botanicals and want to substantiate a natural or organic positioning. Less applicable to a standard hydrocolloid patch.
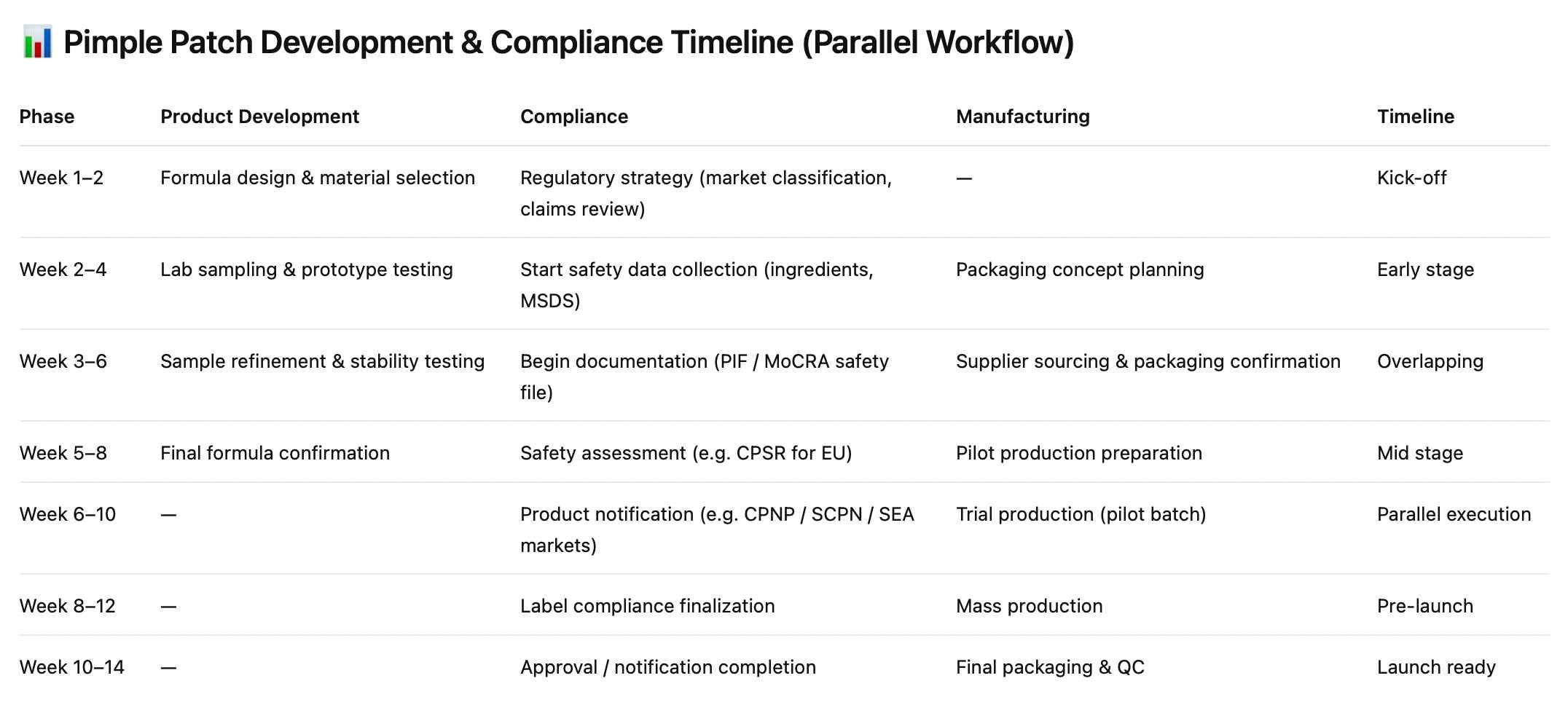
How to Build Your Compliance Timeline
The most common compliance mistake is treating it as a launch task rather than a development task. By the time your product is ready to ship, some registrations should already be months into processing.
Start by mapping your priority markets — where you're selling on day one and where you're targeting in the next 12 months. Then confirm your product classification in each market based on both your formula and your intended label claims. Get this wrong and you're rebuilding from scratch.
Build compliance work in parallel with product development. The EU Responsible Person appointment, CPSR preparation, BPOM registration, and Philippines FDA filing are all slow — they need to start when you finalize your formula, not when you're ready to launch.
Choose a manufacturer who can support this process. That means complete formula documentation with exact concentrations, GMP certification, batch COAs, and regulatory guidance for your target markets. A factory that can't produce these when you ask isn't a partner for a multi-market brand.
And get market-specific support where the stakes are highest. Appoint your EU Responsible Person before a single unit ships to Europe. Engage a local regulatory consultant for Southeast Asian markets where the registration process is opaque without local knowledge.
Quick Reference: Requirements by Market
| Market | What's Required | Timeline |
|---|---|---|
| United States | FDA facility registration + product listing (MoCRA) | Days to weeks |
| European Union | CPNP notification + EU Responsible Person + CPSR | 4–8 weeks |
| United Kingdom | SCPN notification + UK Responsible Person | 2–4 weeks |
| Australia | AICIS compliance + standard cosmetic labeling | Weeks |
| Canada | Post-market notification (within 10 days of first sale) | Days (post-market) |
| Indonesia | BPOM registration | 3–6 months |
| Singapore | HSA notification via PRISM | Weeks |
| Malaysia | NPRA registration via QUEST | Weeks to months |
| Philippines | FDA Philippines registration | 6–12 months |
Frequently Asked Questions
Do I need FDA approval to sell pimple patches in the United States?
No — cosmetic pimple patches don't require pre-market FDA approval. What you do need is FDA facility registration for your manufacturer (mandatory under MoCRA 2022), product listing via the FDA Cosmetics Direct portal, and safety substantiation documentation on file. The exception is if your patch makes acne treatment claims using a recognized OTC active ingredient like salicylic acid — that can trigger OTC drug classification, which has a different and more demanding pathway.
What is the CPNP and do I need it?
The Cosmetic Products Notification Portal (CPNP) is the EU's mandatory pre-market notification system for cosmetics. If you want to sell any cosmetic product — including pimple patches — in the European Union, you must submit a CPNP notification before the product enters the market. The notification must be submitted by an EU Responsible Person; non-EU brands cannot submit it directly.
What is an EU Responsible Person and how do I find one?
An EU Responsible Person is a legal entity based within the EU who takes on formal regulatory responsibility for your product. They submit your CPNP notification, maintain your Product Information File, and act as the point of contact for authorities. Several companies specialize in acting as EU RP for non-EU brands — expect to pay €300–1,000+ per year. Your EU distributor or importer can also take on this role if they're willing.
Does my EU CPNP notification also cover the UK?
No. Since Brexit, the UK operates its own separate system — the SCPN portal (Submit Cosmetic Product Notifications). You need a UK Responsible Person based in Great Britain and a separate SCPN notification. Northern Ireland is an exception: under the Windsor Framework, it still follows EU rules.
Does CE marking apply to pimple patches?
For standard cosmetic pimple patches — hydrocolloid patches sold with cosmetic claims — CE marking does not apply. CE is a medical device designation. It only becomes relevant if your product makes wound healing claims, is intended for post-procedure use, or is otherwise classified as a medical device in the EU.
How long does it take to register a pimple patch in Southeast Asia?
It depends heavily on the country. Singapore (HSA) typically takes a few weeks. Indonesia (BPOM) takes three to six months. The Philippines (FDA Philippines) routinely takes six to twelve months. If you're targeting either Indonesia or the Philippines, begin the registration process as soon as you finalize your formula — not when your product is ready to ship.
Can I use salicylic acid in my pimple patch and still sell it as a cosmetic?
Yes, in most markets — but it depends on your claims and concentration. In the EU, salicylic acid is permitted in leave-on products (patches are leave-on) up to 2.0%, with a mandatory label warning. In the US, salicylic acid at 0.5%–2% is an FDA-recognized OTC active for acne — if you make acne treatment claims, you're in OTC drug territory. If you keep your label copy in cosmetic claim territory and stay within permitted concentrations, you can generally maintain cosmetic classification. Get a regulatory opinion before finalizing your label.
What's the difference between ISO 22716 and GMP certification?
ISO 22716 is the international standard that defines GMP (Good Manufacturing Practice) requirements specifically for cosmetics. When a manufacturer says they're "GMP certified," they typically mean ISO 22716 certified — the two terms are used interchangeably in the cosmetics industry. It's verified through independent audit and is increasingly required by major retailers (Sephora, Boots, Ulta) as a condition of product listing.
Working with VEILTA
Our facilities are ISO 22716 GMP certified, FDA registered, and CPNP compliant. When you work with us, you get full formula documentation, COAs, and regulatory guidance for the US, EU, UK, and Southeast Asian markets — not as extras, but as part of how we work.
If you're still figuring out your compliance requirements for a specific market, our team can help you think through the right approach before you finalize your launch plan.
